Real-world Evidence
Solutions (RWE)
WE ARE YOUR PARTNER FOR
REAL-WORLD
EVIDENCE INSIGHTS
Real-world Evidence
Solutions (RWE)
WE ARE YOUR PARTNER FOR
REAL-WORLD
EVIDENCE INSIGHTS
Our Real-world Evidence Services
Our team has delivered 30+ projects, turning real-world evidence into precise insights for academic research, drug development, and digital health innovation.
Understanding the Current Treatment Landscape
Get a complete picture of prescription patterns, patient subgroups, and treatment paths to see where your product fits and how it can make a difference.
Epidemiology Insights
Understand disease patterns, prevalences, and risk factors. We will provide epidemiological insights to inform public health strategies or optimize your product’s positioning.
External Control Arm Analyses
Strengthen your clinical trial results by comparing real-world outcomes to your clinical trial data. Our results will extend and validate your findings, strengthening regulatory submissions.
Disease Burden Analysis
See the full impact of a disease beyond clinical outcomes. We examine comorbidities, socioeconomic factors, and quality of life to help you address unmet needs.
Comparative Effectiveness Studies
Make data-driven comparisons of treatment options. From RCT to claims data, our analyses reveal which therapies yield the best outcomes, so you can maximize effectiveness.
Long-term Outcomes Monitoring
Evaluate treatment durability over time. With studies spanning one to ten years, we help you understand the long-term impact of your therapies.
Patient-reported Outcome (PRO) Analysis
Know the perceived consequences of treatment switches, from quality of life to fatigue, mood, and satisfaction. Our insights support optimized and patient-centered care.
Patient Satisfaction and Adherence Modeling
Understand why patients stay on or leave treatments. Our models reveal predictive factors for adherence, guiding you to improve engagement.
Post-Marketing Surveillance
Track real-world safety and efficacy. Our regular reporting helps you maintain product confidence, addressing patient feedback and ongoing safety.
Sclerosis
Bowel
Disease
Disease
Gravis
Bowel
Disease
Disease
Sclerosis
Gravis
Bowel
Disease
Disease
Sclerosis
Gravis
Bowel
Disease
Disease
Sclerosis
Gravis
Bowel
Disease
Disease
Sclerosis
Gravis
Bowel
Disease
Disease
Sclerosis
Gravis
Disease Areas
and Countries
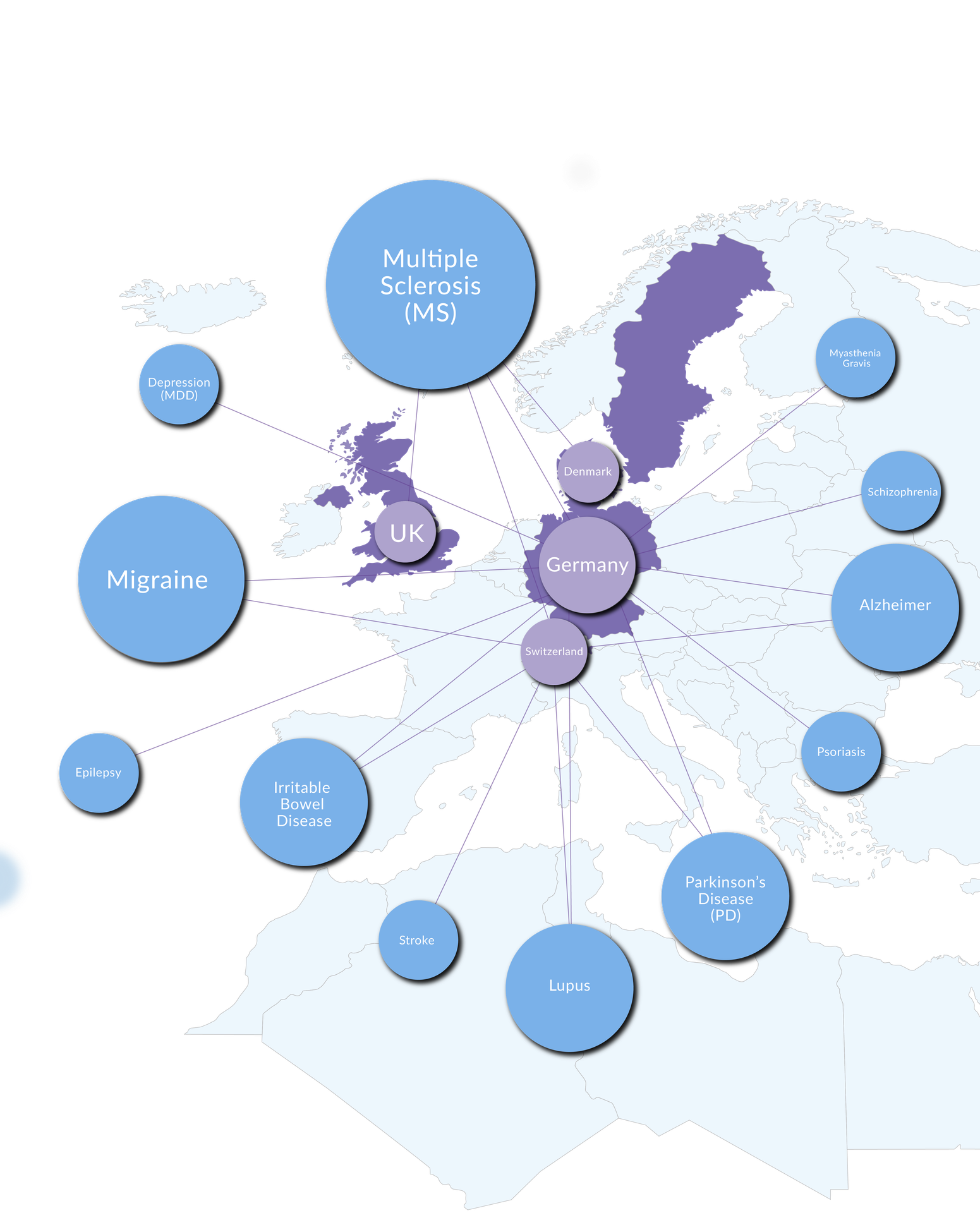
Alzheimer
German RWD
Swiss RWD
Depression (MDD)
German RWD
Epilepsy
German RWD
Irritable Bowel Disease (IBD)
German RWD
Swiss RWD
Lupus (SLE)
German RWD
Swiss RWD
Migraine
German RWD, outpatient
German RWD, clinic-based
Swiss RWD
Multiple Sclerosis (MS)
German RWD
Swiss RWD, medical claims
United Kingdom RWD
Danish RWD
Myasthenia Gravis (MG)
German RWD
Parkinson’s Disease (PD)
German RWD
Swiss RWD
Parkinson’s Disease (PD)
German RWD
Swiss RWD
Psoriasis (PsO)
German RWD
Schizophrenia (SZ)
German RWD
Stroke
Swiss RWD
Case Study: External Control Arms for RCTs with RWD
Real-World Evidence to Enhance Clinical Trial Evidence
The Challenge of Comprehensive Treatment Effects. While randomized controlled trials (RCTs) are the gold standard for evaluating treatments, the health authorities increasingly require data that generalizes and captures real-world effectiveness. Particularly, later-phase trials may suffer from a limited number of comparators, sample sizes, and short follow-up periods. To overcome these issues, external control arms offer a robust and reliable treatment effect evaluation, surpassing the limitations of traditional RCTs and without the untestable assumptions of network meta-analyses.
The Approach to Broaden Treatment Effects. To empirically address critiques about RCTs, we use real-world data (RWE) to generate quantitative effects of the treatments across multiple comparators, in larger samples in actual clinical practice. How? Keeping the RCT as the gold standard we carefully select a suitable control group for the RCT from a large real-world database, aligning the RW population to the RCT population, thoroughly validating the approach, and deploying advanced methods like propensity matching, weighting, and target-trial emulation. Through this, we can provide additional comparator arms for RCTs from RWD while minimizing selection bias and controlling.
Benefits
of the Rewoso
approach
GreaterRelevance
Show the effects of treatments across diverse patients and settings by using real-world data.
Accelerated Development
Accelerate drug development timelines by leveraging existing, propensity-matched real-world cohorts.
Factual Decision-Making
Get data-driven information for important decisions and the optimization of treatment strategies.
Cost-Effective Research
Real-world studies often cost less than traditional trials by reusing existing data sources.
Beyond Network Meta-Analyses
External control arms yield individual-level comparisons, with fewer assumptions than meta-analyses.
Benefits of the Rewoso approach
Greater Relevance
Show the effects of treatments across diverse patients and settings by using real-world data.
Accelerated Development
Accelerate drug development timelines by leveraging existing, propensity-matched real-world cohorts.
Factual Decision-Making
Get data-driven information for important decisions and the optimization of treatment strategies.
Cost-Effective Research
Real-world studies often cost less than traditional trials by reusing existing data sources.
Beyond Network Meta-Analyses
External control arms yield individual-level comparisons, with fewer assumptions than meta-analyses.















